Researchers in Oman have developed a simple and sensitive test for the rapid diagnosis of COVID-19.
The research project is conducted by a team led by Dr Haytham Ali from Sultan Qaboos University (SQU).
It is funded within the COVID-19 Research Programme, one of the research projects funded by the Ministry of Higher Education, Research and Innovation (MoHERI).
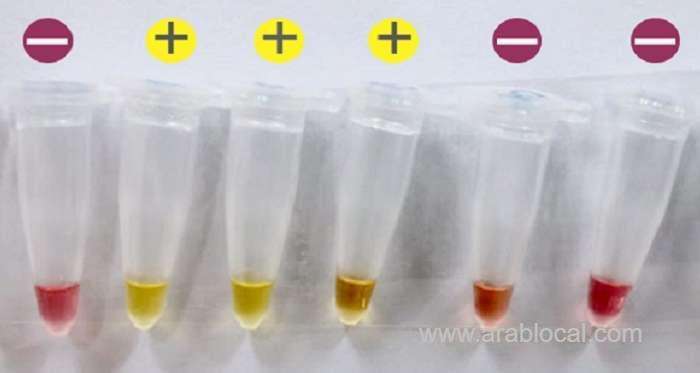
The test is a ready-to-use, rapid and sensitive colorimetric Reverse Transcription Loop-Mediated Isothermal Amplification (RT-LAMP) based assay for the diagnosis of COVID-19 viral infection.
The research idea is derived from the fact that a rapid and sensitive COVID-19 test is needed for the prevention and control of the current COVID-19 pandemic spread.
“The developed colorimetric assay is highly sensitive and was able to detect as low as 80 viral genome copies with possible improvement through further adjustments,” Dr. Haytham Ali, the Principal Investigator of the research project, said.
Three primer sets were designed by Dr Timothy Holton from SQU (co-principal investigator) based on the complete genome sequences of forty one SARS-CoV-2 isolates from Oman, targeting the Spike protein gene and the M gene. The primer set (CoV_S23258) was found to be the most sensitive and specific among the three designed sets.
The RT-LAMP assay was validated by testing 145 COVID-19 clinical samples obtained from SQU Hospital with a sensitivity of 96.9 per cent and specificity of 94.7 per cent when compared to the validated Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR) assay.
The RT-LAMP assay specificity was tested against SARS-CoV Frankfurt-1 RNA virus and avian corona viruses as they tested negative with the developed assay.
The research team examined the possibility of directly using saliva samples spiked with SARS-CoV-2 RNA that gave preliminary excellent results with the developed assay.
Currently, Dr Haytham’s team is trying to convert the test into a lyophilised (freeze dried) ready to use assay, where water and template need to be added to run the test. Results can be obtained by reading the colour change from red to yellow.
“Once we validate the lyophilized assay, we will proceed of implementing the assay as a point of care test in clinics, airports, and other places,” Dr Haytham added.
The assay can be utilised at its current status as a screening assay with the advantages of being simpler, quicker (20-30 minutes) and cheaper than the qRT-PCR.
SOURCE: TIMESOFOMAN